
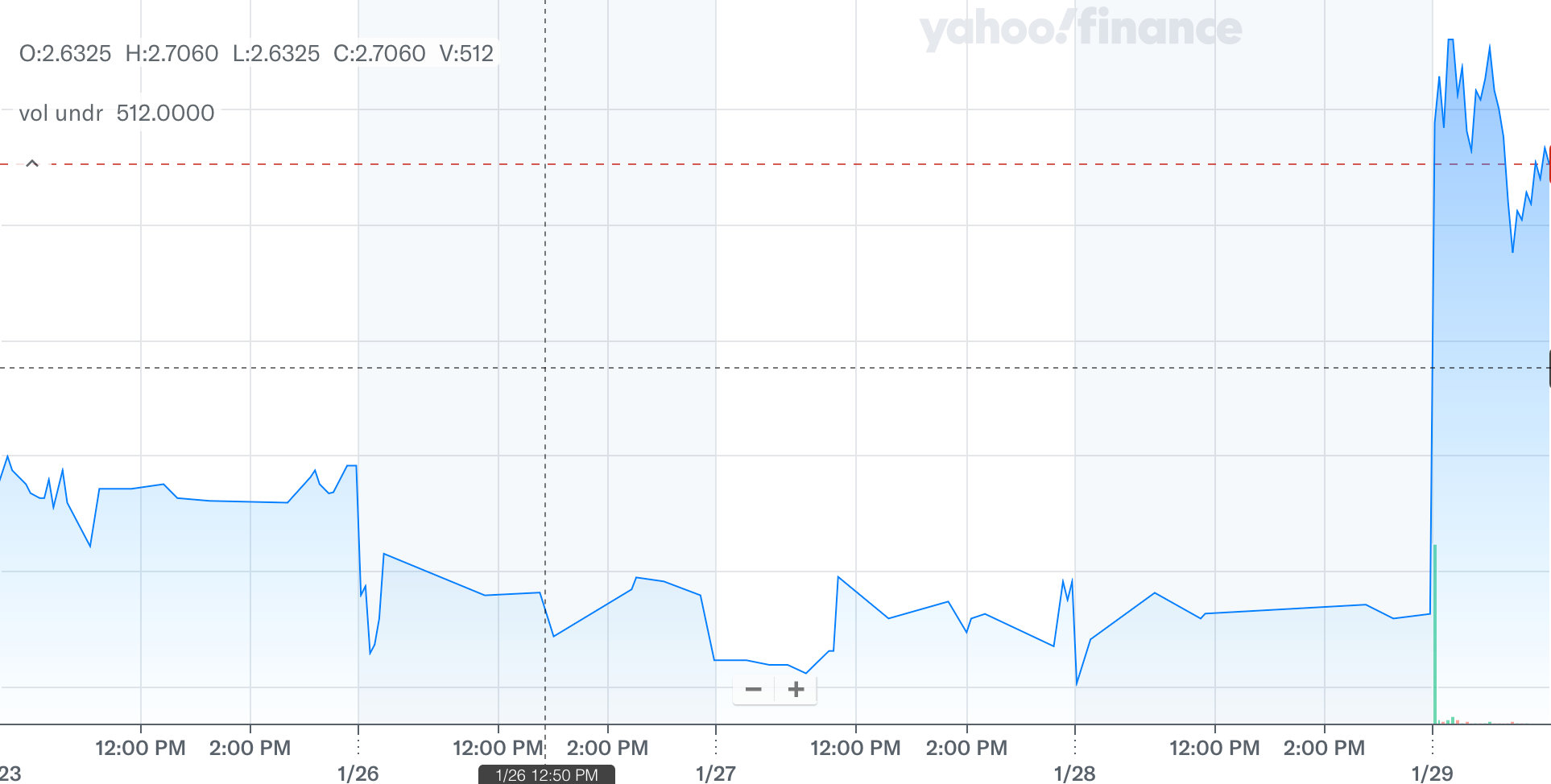
Serina Therapeutics (NYSE: SER) surged sharply today after the company announced that the U.S. Food and Drug Administration (FDA) cleared its Investigational New Drug (IND) application for its lead therapy candidate, SER-252, which is being developed to treat advanced Parkinson’s disease.
In simple terms, SER-252 is designed to deliver a Parkinson’s medication slowly and continuously, instead of in short bursts. It’s based on a drug called apomorphine, which helps replace missing dopamine in the brain, but it uses a special polymer technology to release the medicine at a steady rate, usually through a small under-the-skin pump.
Now the FDA clearance enables Serina to proceed with site-level start-up and regulatory activities that support the initiation of a Phase 1b registrational clinical study under a New Drug Application pathway, which could streamline development toward an eventual NDA submission.
This regulatory alignment is widely viewed as a major operational de-risking event for the company, prompting heavy trading volume and a double-digit percentage jump in the stock driven by renewed investor optimism around clinical progress.
To be sure, the FDA’s clearance of SER-252’s IND marks one of the most significant catalysts for Serina in recent quarters, especially given previous regulatory challenges and communications with the agency.
With the registrational study now enabled, Serina has a clearer path to begin generating meaningful human clinical data in Parkinson’s patients.
Wall Street coverage of Serina remains limited but optimistic.
Recent consensus estimates from analysts place the average 12-month price target at around $13.00, implying a gain of more than 225%.







.webp)