
The market opened up lower this morning, following President Trump’s speech last night.
But that didn’t stop Cocrystal Pharma (NASDAQ: COCP) from getting a nice bump after announcing that the FDA had granted it Fast Track designation for the company’s oral, direct-acting protease inhibitor, CDI-988.
This is the first oral antiviral candidate being developed for the treatment and prophylaxis of norovirus infection, which is a highly contagious virus responsible for roughly 685 million cases globally each year and tens of billions in economic impact.
Here’s how the market responded to the news …
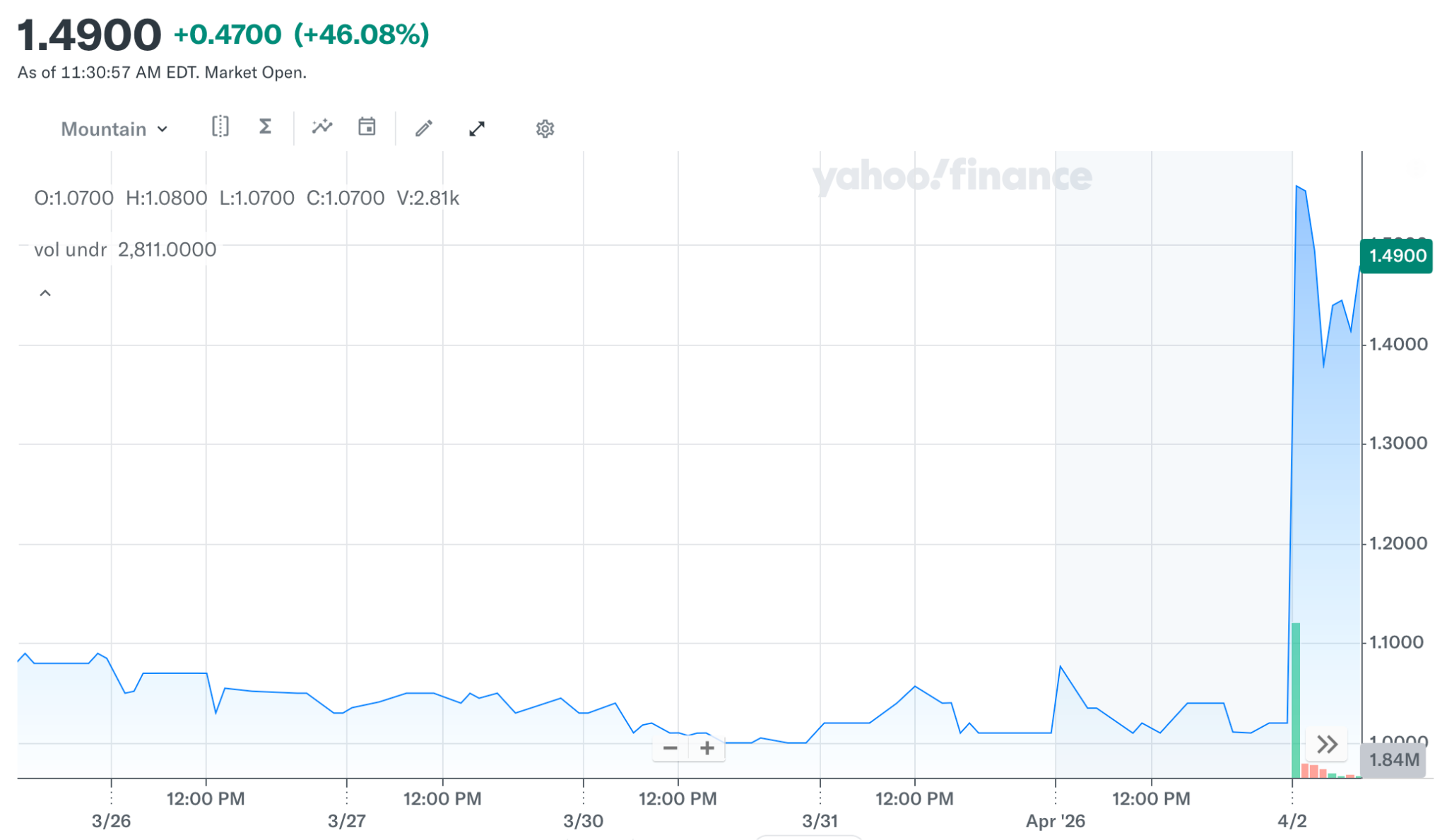
To clarify, Fast Track designation doesn’t mean approval, and it doesn’t eliminate risk. But it does mean the FDA sees this as a serious unmet need and is willing to move faster than usual. As a result, Cocrystal gets a more direct line into the regulatory process.
That includes more frequent communication with regulators, the ability to submit data on a rolling basis, and potential eligibility for priority review down the line.
Why this drug matters
CDI-988 isn’t just another incremental therapy. It’s being positioned as the first oral antiviral candidate designed to both treat and prevent norovirus infections. That’s a big deal because there’s currently no approved antiviral treatment for norovirus.
To be sure, the program is still early. CDI-988 is currently in a Phase 1b challenge study, meaning:
- Small patient population
- Early-stage safety and efficacy signals
- Plenty of room for things to go wrong
This is not a late-stage, de-risked asset. But it’s a promising idea moving through the earliest real-world testing phase.
With Fast Track designation in place, the burden now shifts back to the company. Because from here, nothing matters more than the data.
If CDI-988 delivers in clinical trials, this Fast Track designation becomes a launchpad. If it doesn’t, it’s just another early-stage biotech story that never made it out of the gate.
Let’s hope it’s the former.







.webp)