
Psychedelics stocks are on fire this morning.
AtaiBeckley (NASDAQ: ATAI), Compass Pathways (NASDAQ: CMPS), and Definium Therapeutics (NASDAQ: DFTX) are all up big following President Trump’s executive order to accelerate access to psychedelic drug treatments.
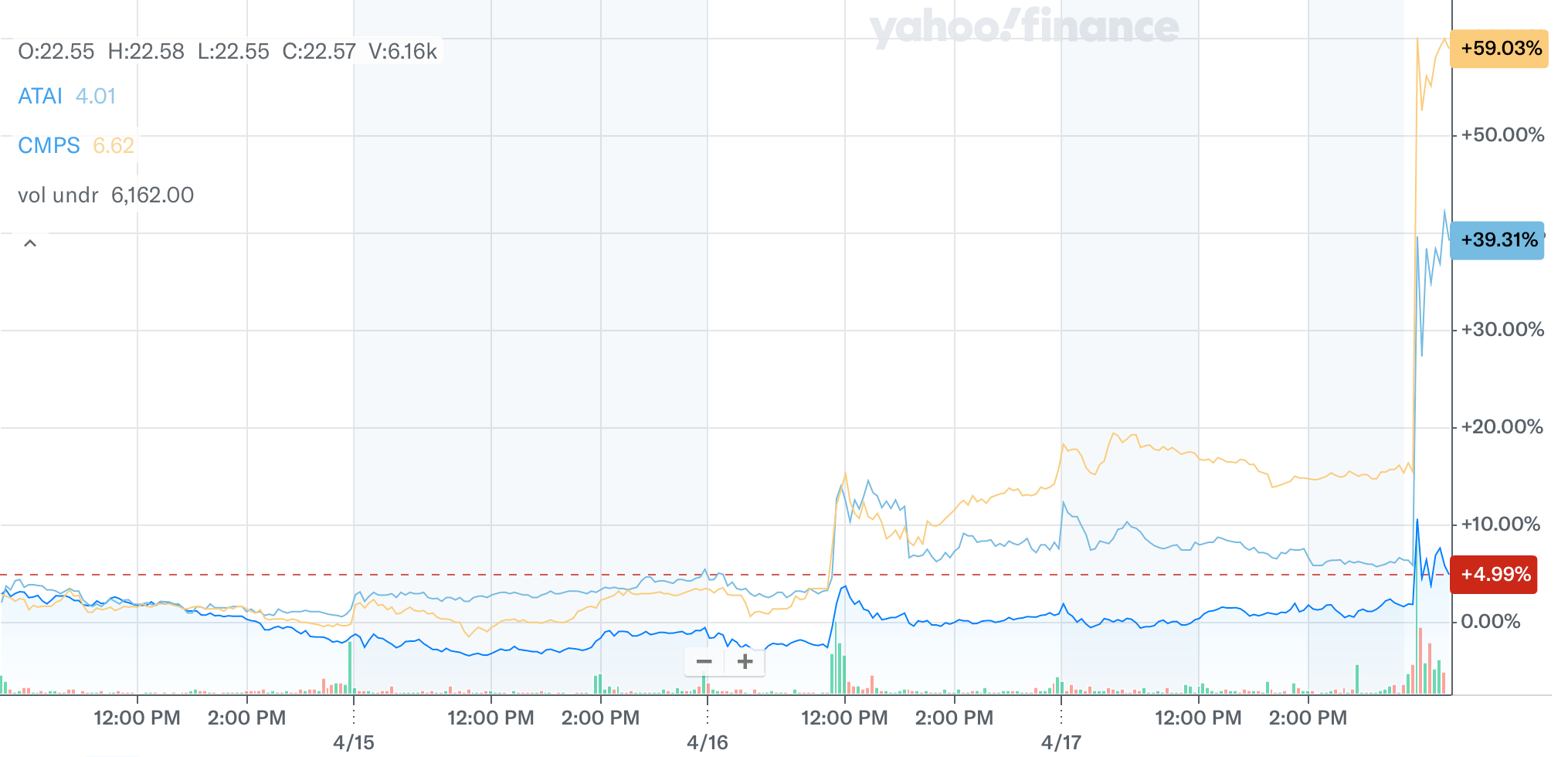
The order essentially directs the FDA to speed up reviews of psychedelic drugs, particularly those already designated as “breakthrough therapies.”
That matters because the review window could shrink from the typical 6 to 10 months down to as little as 1 to 2 months in some cases. Shorter timelines mean faster approvals, lower capital burn, reduced dilution risk, and earlier revenue potential.
For a sector that has already made great strides without federal policy support, this completely changes the narrative.
This Is a Regulatory Signal, Not a Scientific Breakthrough
To be sure, nothing changed on the science side since Saturday, when the executive order was signed.
No new Phase 3 data, no new approvals, no new clinical outcomes.
What changed is the probability distribution of outcomes.
The government is now:
- Prioritizing these therapies
- Allocating funding (roughly $50 million for research)
- Creating mechanisms to accelerate access and review
That lowers regulatory risk, which is one of the primary discount factors in early-stage biotech.
The Stocks That Actually Benefit
The market reaction looks broad on the surface, but the impact of this executive order is highly selective. The benefit focuses on where time compression actually matters: late-stage clinical programs with a clear regulatory path.
Start with Compass Pathways. Its lead asset, COMP360, is already in Phase 3 trials for treatment-resistant depression. That puts it in a narrow window where accelerated FDA review can materially change valuation.
If timelines compress, Compass moves from a “multi-year story” to a near-term regulatory event. In biotech (which is really where psychedelics fit into the equation), that shift alone can drive multiple expansions because capital is no longer locked up waiting on uncertain timing.
The same dynamic applies to AtaiBeckley with its lead asset, BPL-003, which is designed specifically for scalability. This, as a result of shorter treatment durations and clinic-friendly protocols.
This is important because the FDA isn’t just approving molecules, it’s implicitly evaluating delivery models. A therapy that fits into a practical clinical workflow has a clearer commercialization path, which makes accelerated review more meaningful.
Then you have Definium Therapeutics, which is advancing LSD-based therapies for generalized anxiety disorder and ADHD, with late-stage trials underway.
Again, the key variable is proximity to a decision point. If you’re within striking distance of Phase 3 completion, regulatory acceleration directly impacts expected return timelines.
Overall, Trump’s executive order isn’t about hype. It’s about math.
If you reduce regulatory timelines, increase federal support, and signal institutional acceptance, you change the economics of the entire sector. That’s what the market is pricing in today.
And right now, it’s moving faster than the fundamentals, because policy just moved first.








.webp)